  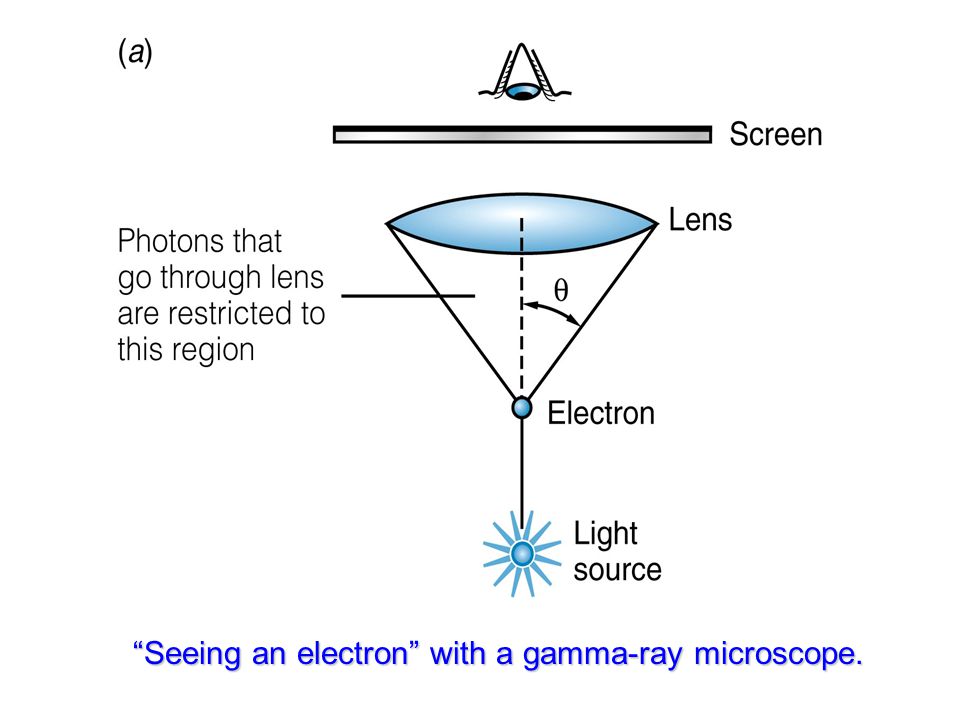 
Schrödinger postulated his equation in 1925 and published. But the formal inequality we know with the standard deviations and h bar was derived in 1928 by someone else. Heisenberg published it in his paper in 1927 as: pq h. This reflected photon causes a change in the path of the electron. Explain why the Heisenberg Uncertainty Principle helps to justify the fact that a wave function can only predict the probable location of an electron and not its exact location. Limited by Heisenberg's uncertainty principle. Heisenberg first introduced his uncertainty principle in 1927, after the Schrödinger equation had been published. Heisenberg Uncertainty Principle: The observation of an electron with a microscope requires reflection of a photon off of the electron. Explain what the Heisenberg Uncertainty Principle means in terms of the position and momentum of an electron. Packets with narrower position and momentum widths, but this is ultimately These difficulties can be mitigated by employing wave Louis de Broglies particle-wave duality particles with mass, Max Plancks and Albert Einsteins work with quantum physics, Neil Bohrs earlier model of the atom, Werner Heisenbergs. As a consequence of dual nature of matter, Heisenberg, in 1927 gave a principle about the uncertainties in simultaneous measurement of position and momentum. Practice and whose verification may require rather long integration times.įurthermore, in highly sensitive configurations gravity gradients lead to aĭrastic loss of contrast. Indeed, the relative initial position and velocity for the two species need toīe controlled with extremely high accuracy, which can be rather demanding in Highest possible sensitivities, gravity gradients pose a serious challenge. In Niels Bohrs theory of the atom, electrons absorb and emit radiation of fixed wavelengths when jumping between fixed orbits around a nucleus. Download a PDF of the paper titled Circumventing Heisenberg's uncertainty principle in atom interferometry tests of the equivalence principle, by Albert Roura Download PDF Abstract: Atom interferometry tests of universality of free fall based on theĭifferential measurement of two different atomic species provide a usefulĬomplement to those based on macroscopic masses. 
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
